No products in the cart.
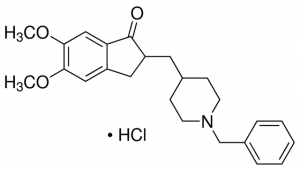
donepezil hydrochloride
| Chemistry data | |
| formlula | C24H29NO3. 2HCl |
| Molecular Weight | 379.492 g/mol |
| CAS Number: 120011-70-3 | |
Category: No categorization
Therapeutic category: Central acetylcholinesterase inhibitor
Dosage forms: tablets
Indications: The use of this drug in mild to moderate and severe stages of Alzheimer’s disease has been confirmed. It has no effect on the process of neurodegeneration and only improves the symptoms of cognition, mental activity, memory, attention, social communication, reason, speech and daily activity of the patient. Sometimes taking dopezil with memantine in the treatment of Alzheimer’s disease is superior to taking dopezil alone.
Mechanism of action: Blocks the enzyme acetylcholinesterase. Donepsil is a non-competitive, reversible specific inhibitor of acetylcholinesterase that increases extracellular acetylcholine.
Drug metabolism: The bioavailability of the drug is 100%. Donepsil is well absorbed from the gastrointestinal tract and its maximum plasma concentration is reached 3-4 hours after administration. 96% of dopezil binds to plasma proteins, especially albumin. Donepezil is metabolized in the liver by cytochrome P450, 3A4, and 2D6 isoenzymes to four major metabolites, two of which are active. Steady-state plasma concentrations are reached 3 weeks after the start of treatment. Excretion of the drug and its metabolites is predominantly in the urine (79%) and to a lesser extent (21%) in the feces. The elimination half-life of dopezil is 70 hours
Side effects: nausea, vomiting, diarrhea, fatigue, insomnia, dizziness, anorexia, weight loss, gastric and duodenal ulcers, muscle cramps, tremor, conduction disorders of the heart, bradycardia, extrapyramidal effects of depression, abnormal skin behavior, zinc Skin discoloration and liver disorders, Muscle cramps, Sweating, Urinary tract infection and Respiratory tract infection
Be the first to review “donepezil hydrochloride” Cancel reply
Related products
No categorization
No categorization
No categorization
No categorization
No categorization
No categorization








Reviews
There are no reviews yet.